


1 / 5










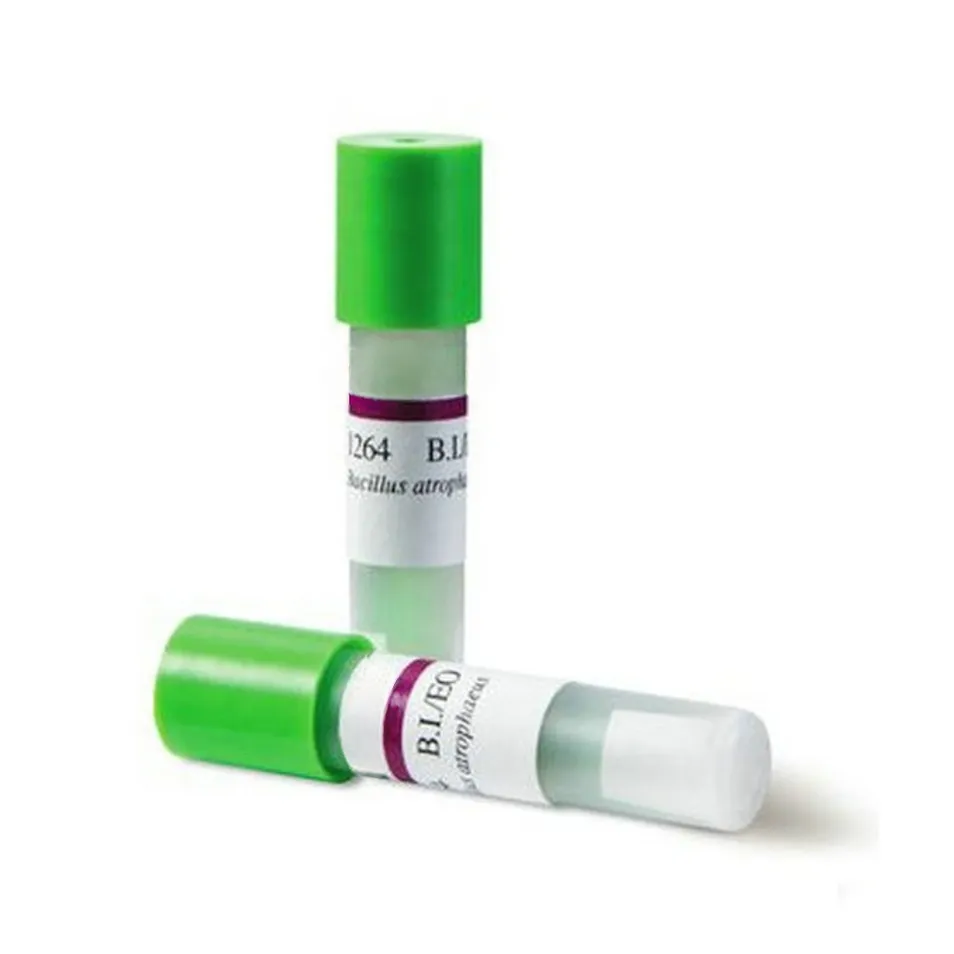
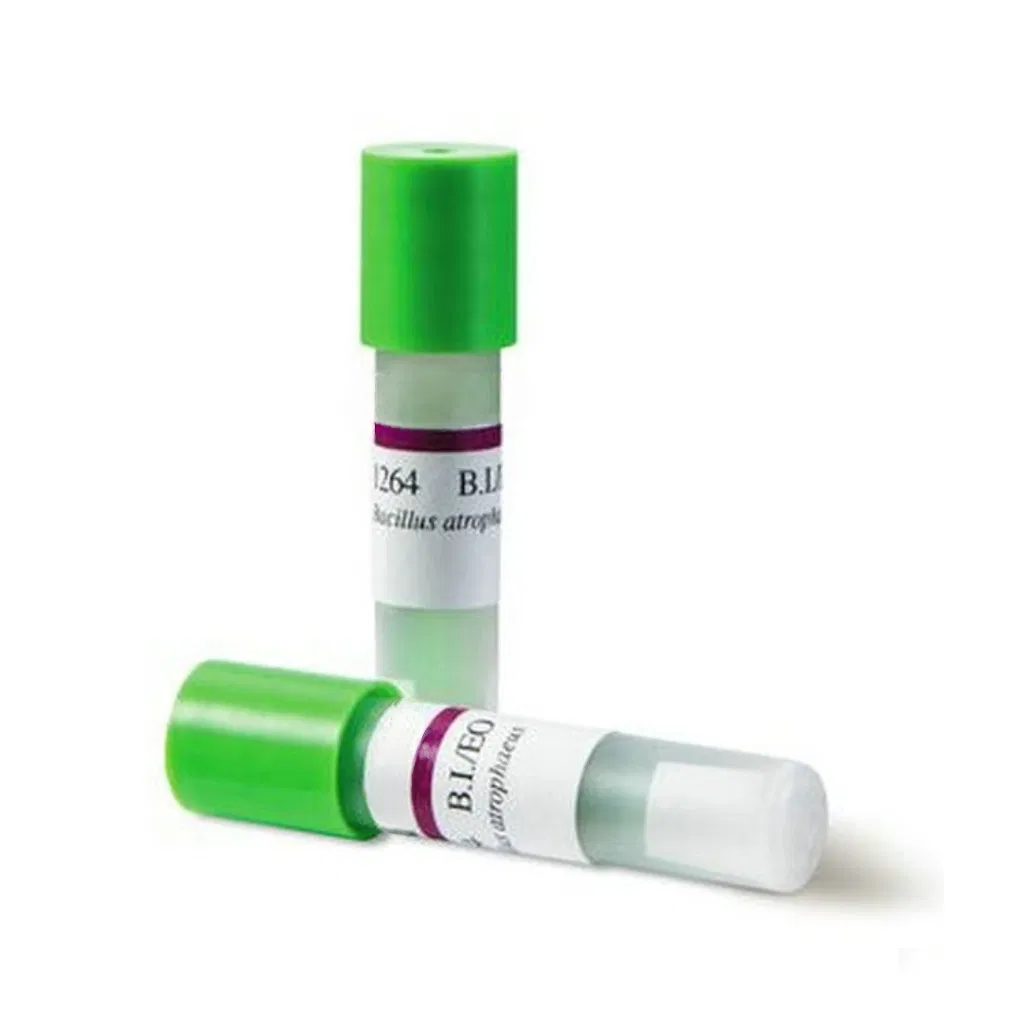
Purpose: For biological monitoring of ethylene oxide sterilizers.
Mechanism: The spores are those of Bacillus subtilis var. niger. Tested with a standard resistance meter: the survival time is 15 minutes or more, and the killing time is 60 minutes or less.