Basic Specifications
Model NO.FST-SIZE
Electric Tension220~380V
LCD DisplayWith LCD Display
MaterialStainless Steel
AlarmsUltra High Temperature Alarm
RecordingInfo Recording & Printing
Doors TypePneumatic Sliding/Revol Door
Capacity Range1cbm to 100cbm
Product Introduction
⚙
Our products include the overall facilities of EO sterilization chamber, preconditioning chamber, degassing chamber, and EO waste gas treatment apparatus.
📋
All products meet the stringent requirements of ISO9001:2008 and ENISO134585:2003/AC2009.
💡
Innovative hot air heating system technology with utility model patent certification. Leading the ethylene oxide sterilization industry.
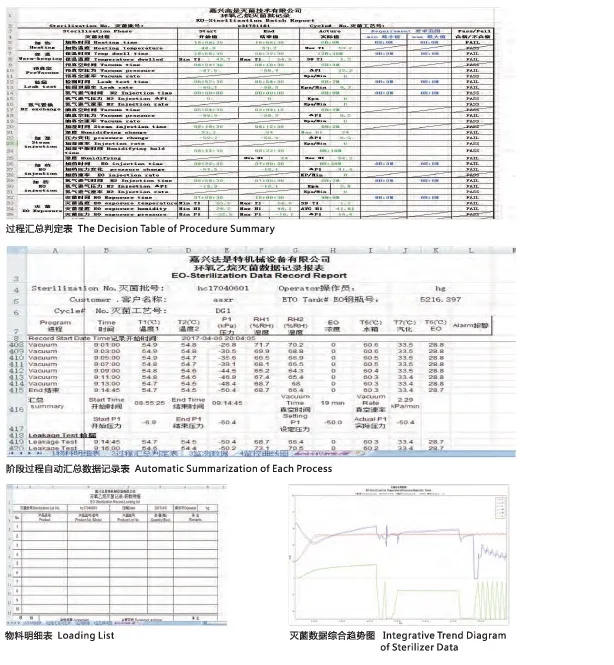
Sterilization Cycle Process
- Loading: Goods placed in sterilizer and door locked securely.
- Temperature Adjusting: Reaching 50ºC (approx. 1-2 hours).
- Pre-Vacuum: Air removal (6-30 minutes).
- Leak Test: Checking tightness to ensure zero gas leakage.
- Pre-humidification: Adjusting to 55%RH humidity (1-1.5 hours).
- EO Injection: Injecting Ethylene Oxide gas (20-30 minutes).
- Sterilizing: Maintaining conditions for 4-8 hours.
- Gas Clearing: Residue removal via vacuum (at least 3 cycles).
- Unloading: Safety unlock and cart removal.
Efficiency: The sterilization chamber can process two batches of cargo within 24 hours.
Frequently Asked Questions
Q1: What is the available capacity for the EO sterilization chambers?
A: We offer a wide range of capacities from 1 cubic meter (1cbm) up to 100 cubic meters (100cbm), accommodating 1 to 13 pallets.
Q2: Which international standards do your sterilizers comply with?
A: Our products are designed to meet ISO9001:2008 and ENISO134585:2003/AC2009 standards, ensuring high quality and safety for medical use.
Q3: How many batches can be sterilized in a day?
A: The E.O. gas sterilizer is highly efficient and is capable of processing two full batches of cargo within a 24-hour period.
Q4: What material is used for the sterilization chamber construction?
A: The chambers are constructed using high-grade stainless steel, specifically Grade 304 or 316, for maximum durability and corrosion resistance.
Q5: Do you provide support for international customers?
A: Yes, we provide comprehensive after-sales support including installation training and have engineers available to service machinery overseas.
Q6: Is the control software compliant with medical data regulations?
A: Yes, our control software is qualified under FDA 21CFR part 11, featuring data storage, graphic recording, and safety password administration.