The E.O. Gas Sterilization Process
1
Loading: Securely place goods and lock the chamber.
2
Temperature Adjustment: Reaching 50ºC (approx. 1-2 hours).
3
Pre-Vacuum: Removing air from the chamber (6-30 mins).
4
Leak Test: Ensuring system tightness for gas safety.
5
Pre-humidification: Adjusting humidity to 55%RH (1-1.5 hours).
6
Gas Injection: Injecting EO gas into the chamber (20-30 mins).
7
Sterilizing Stage: Maintaining constant conditions for 4-8 hours.
8
Gas Removal: Clearing residue via vacuum and air blowing (approx. 1 hour).
9
Unloading: Unlocking door and retrieving sterilized goods.
Capacity Efficiency: Our sterilizer chamber can process two batches of cargo within 24 hours.
Frequently Asked Questions (FAQ)
Q1: What is the capacity range of your EO sterilizers?
A: We offer a wide range of chamber capacities from 1 cubic meter (cbm) up to 100 cubic meters, suitable for various industrial needs.
Q2: What materials are used to ensure the durability of the chamber?
A: The chambers are constructed using high-grade stainless steel (304 or 316) to ensure corrosion resistance and long-term durability.
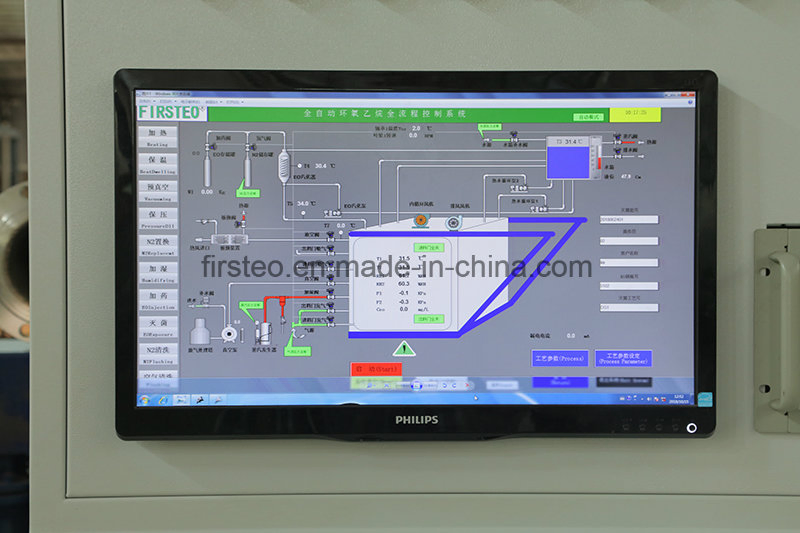
Q3: Does the software comply with international medical standards?
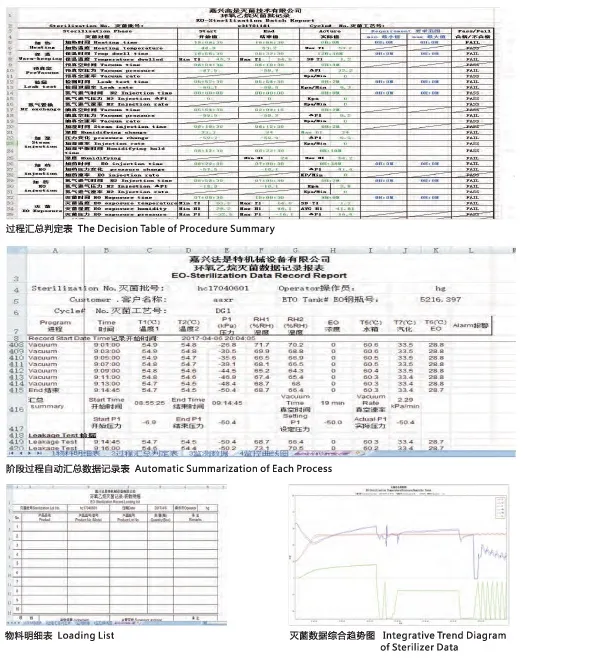
A: Yes, our control software is designed for automatic and manual operation, featuring data storage and graphic recording of all critical parameters. Optional FDA 21CFR Part 11 compliant software is also available.
Q4: How efficient is the sterilization cycle?
A: Our systems are highly efficient, capable of processing two full batches of cargo within a 24-hour window, depending on the material requirements.
Q5: Do you provide support for international customers?
A: Absolutely. We provide comprehensive training, and our engineers are available for overseas service, installation, and long-term maintenance support.
Q6: What safety features are integrated into the system?
A: The system includes ultra-high temperature alarms, pneumatic safety door locks, pressure/leak testing stages, and specialized waste gas treatment units to ensure total operational safety.