














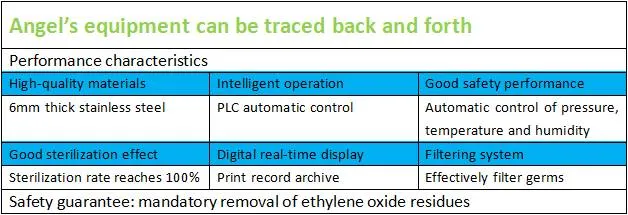
Aggregated professionals in the field of medical equipment and concentrated technical forces have achieved a series of results in ethylene oxide sterilizers. Combined with the 5G intelligent AI platform to realize the intelligent operation of the equipment, the simplicity, safety, and sterilization effect exceed requirements. This equipment represents a large-scale medical instrument solution for sterilization and disinfection devices.

The ethylene oxide molecule generates an alkylation reaction with carboxyl, amino, and sulfhydryl groups in bacterial cell proteins. This destroys metabolic reactions and effectively kills various microorganisms, including spores, mycobacterium tuberculosis, viruses, and fungi.
Low-temperature sterilization method suitable for cotton, plastic films, and other sensitive materials.



| Model | HTAJ-2CBM | HTAJ-6CBM | HTAJ-10CBM | HTAJ-20CBM |
|---|---|---|---|---|
| Rated Power | 9KW | 18KW | 24KW | 35KW |
| Liner Size (mm) | 1500*1000*1400 | 3000*1200*1700 | 4000*1500*1800 | 6000*1750*1950 |
| Working Temp | 30ºC ~ 50ºC (Adjustable) | |||
| Working Pressure | -60Kpa ~ 0 | |||
| Weight | 1500kg | 3200kg | 4600kg | 8000kg |



1. Space: Recommended independent space (>20 cubic meters) with excellent ventilation and explosion-proof fittings.
2. Utilities: DN40 outlet hose for sewage, DN20 for overflow, and standard tap water inlet hose.
3. Power: Three-phase five-wire power distribution box with copper core cables.
4. Air: Air compressor (above 4KG) required for each unit.
